|
(His mathematician brother Harald, a former Olympic soccer player, would go on to open the university’s mathematical institute next door nine years later.) In 1965 the university renamed the facility the Niels Bohr Institute, and today more than 1000 staff and students work and study there. He was granted approval, and the institute opened in 1921 with Bohr serving as director. Soon after, he began pushing for a new institute for his field, which would allow researchers from all over the world to collaborate with Danish scientists at a state-of-the-art facility. HE FOUNDED COPENHAGEN’S INSTITUTE FOR THEORETICAL PHYSICS.īased on his atomic research, the University of Copenhagen hired Bohr as a professor of theoretical physics in 1916 when he was just 31 years old. 
Bohr eventually tweaked Rutherford’s model by explaining that the electrons orbiting a positively charged nucleus can jump between energy levels, which stabilizes the atoms. By the laws of physics, the orbiting electrons should eventually crash into the nucleus and destabilize the atom. The pair studied the structure of the atom, and Bohr determined Rutherford’s model must not be entirely correct. This became the foundation of his work with Bohr. Because some of the particles bounced back instead of going through the gold, he determined the majority of the atom’s mass must be within a small, central nucleus, with the electrons orbiting around it. Rutherford had discovered the atomic nucleus through an experiment in which he shot alpha particles at a thin sheet of gold foil. BOHR NAILED THE TRUE STRUCTURE OF AN ATOM IN 1913.Īfter finding his work at odds with Thomson’s, Bohr joined the Manchester University lab of Ernest Rutherford, who had also studied under Thomson. Bohr would later contradict the “plum pudding” structure with his atomic model. This led Thomson to propose the “ plum pudding” structure of atoms, in which negatively charged electrons are embedded in a sphere of positively charged matter, like raisins in a English pudding. But he eventually determined that the rays were actually particles even smaller than the atom by showing that they could be deflected by electricity. Thomson had turned his attention to cathode rays, which were then believed to be part of the ether-a theoretical, weightless substance found everywhere in the universe. Thomson, who had discovered the electron in 1897. BOHR DISAGREED WITH HIS PROFESSOR’S “PLUM PUDDING” THEORY.Īfter graduating, Bohr continued his studies at Cambridge University under J.J. He would, however, go on to earn a master’s degree in 1909 and a doctorate in 1911 in physics. Bohr studied other fields, including inorganic chemistry, perhaps less successfully: He earned a reputation for causing explosions in the lab, and eventually broke a record amount of glass at the school. While he initially studied mathematics and philosophy, he won a physics competition sponsored by the Royal Danish Academy of Sciences, and he soon changed his major to physics. HE SET OFF EXPLOSIONS IN HIS UNIVERSITY'S CHEMISTRY LAB.īohr began his university studies in 1903 at the same institution that employed his father, the University of Copenhagen. Later in life, he penned a number of philosophical writings on physics, having overcome his youthful aversion to exposition. He excelled in the majority of his studies, and he graduated first in his class. There we saw ships land and leave."īut by secondary school, he was correcting errors that he discovered in his physics textbooks.  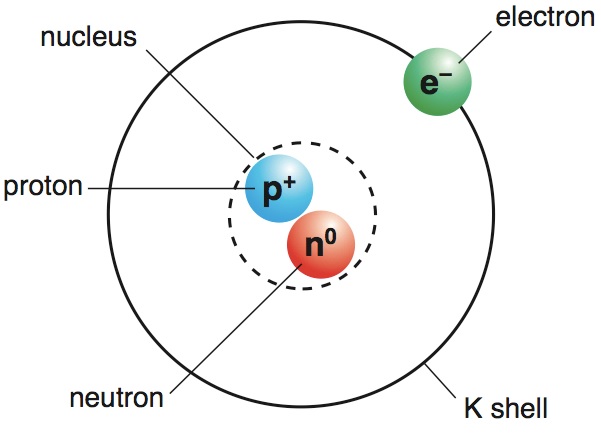
According to the Niels Bohr Institute at the University of Copenhagen, he once turned in an essay that contained just two sentences: "A trip in the harbor: My brother and I went for a walk in the harbor. NIELS BOHR WAS A STELLAR STUDENT BUT A MEDIOCRE WRITER.īohr enrolled at the Gammelholm Latin School at age 7 and did well in all of his classes except for composition. Though he never won, Christian Bohr was nominated for the Nobel Prize by one colleague in 1907 and by two in 1908, all for his research on the physiology of respiration. Young Niels and his two siblings often listened in, which likely inspired the young student’s future studies. His father Christian was a physiology professor at the University of Copenhagen, and he often hosted fellow scientists at his home for lively discussions. Niels Bohr, born in Copenhagen in 1885, was brought up in a family that valued science. HIS FATHER WAS NOMINATED FOR NOBEL PRIZES THREE TIMES IN TWO YEARS. Here are 13 things you might not have known about Bohr. In the early 20th century, Danish physicist Niels Bohr discovered the basic atomic structure-a positively charged nucleus surrounded by orbiting electrons-which laid the groundwork for how we understand atoms today. Quantum physics might not be the most approachable topic, but there’s a good chance you’ve heard of some of its elemental parts, like atoms.
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
